This text is adapted from Openstax, Chemistry 2e, Section 6.5: Periodic Variations in Element Properties. Potassium superoxide is a yellow solid that decomposes at 560 ☌. Superoxides are characterized by the presence of O 2 −. The alkali metals such as potassium, rubidium, and cesium also produce superoxides. The peroxide ion has a single oxygen-oxygen covalent bond and is a powerful hydrogen ion acceptor, making the peroxides of the alkali metals strong bases. Lithium reacts with oxygen to give lithium oxide, while other alkali metals like sodium form oxides and peroxides. Both of these reactions become more exothermic for the heavier alkali metals due to their lower ionization energies.Įxposure to air, as they react with oxygen to form oxides, reduces the metallic luster of the alkali metals. The heat produced during the reaction can ignite the hydrogen gas released, leading to an explosion. The alkali metals also react violently with water, also, and hydrogen gas and an alkali metal hydroxide are produced. The reaction of sodium and chlorine to produce sodium chloride is exothermic. The alkali metals react vigorously with nonmetals, such as halogens. Their ability to lose an electron readily makes them highly reactive and excellent reducing agents. Thus, alkali metals have a great tendency to give away this electron and have low first ionization energies, which decrease down the group. The loss of this electron produces a cation with the noble gas configuration. The electron configuration of these elements shows that the valence shell has only one electron (Table 1). Thus, the outer electrons get farther from the nucleus, and the atomic radius increases from lithium to cesium. Moving down a group, the principal quantum number, n, increases by one for each element. Each element can usually be classified as a metal or a non-metal based on their general Physical and Chemical Properties. Table 1: Properties of the alkali metals Element Alkali Metals are used as a term in Periodic Chemistry when classifying the chemical elements. The melting points of the alkali metals are unusually low for metals and decrease going down the group, while the density increases going down the group with the exception of potassium (Table 1). They are malleable, ductile, and good conductors of heat and electricity.  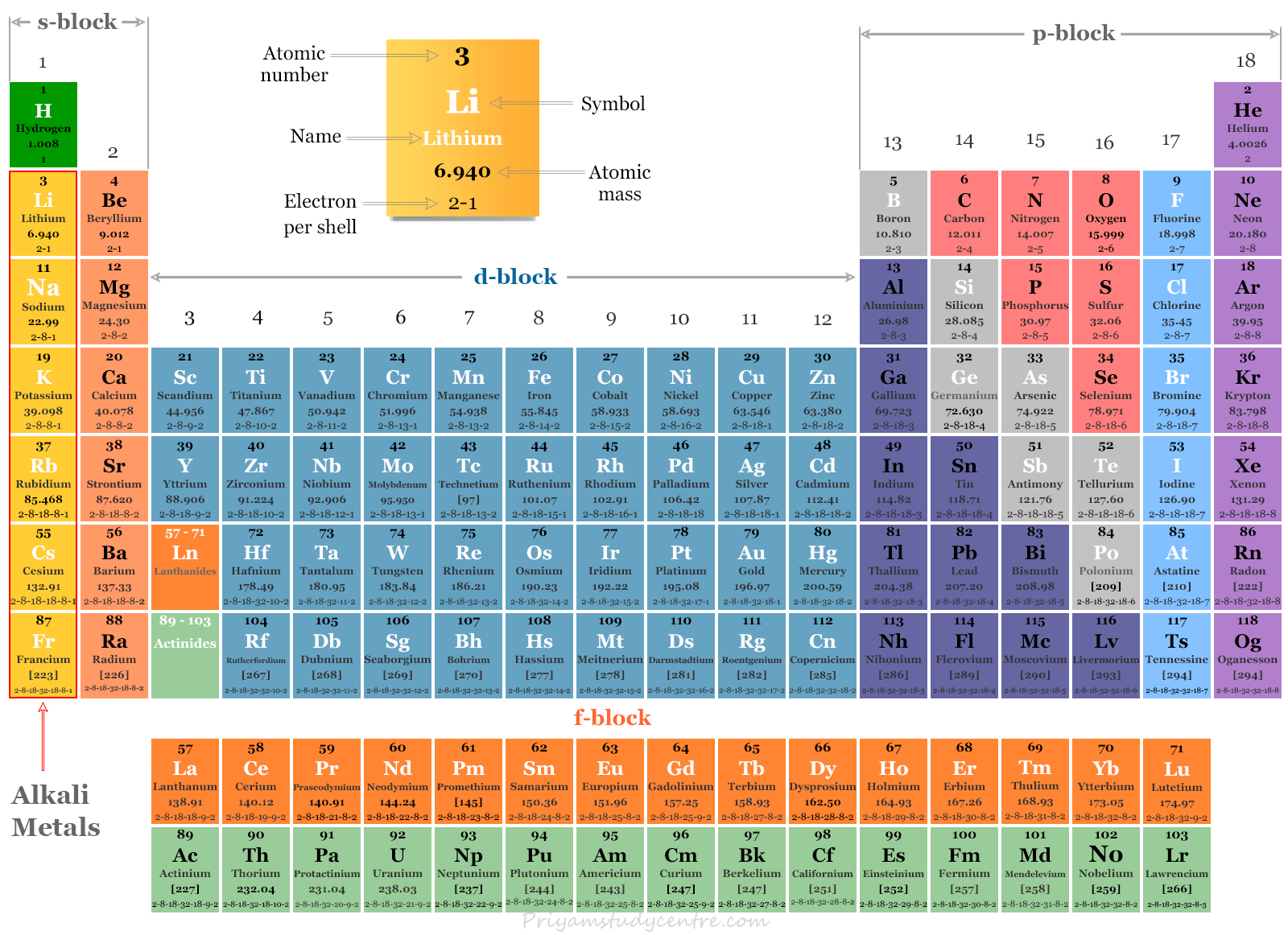
Group 1 elements are soft and shiny metallic solids. The emission spectrum of each element is unique and used to identify the element qualitatively. When the excited electrons return to the ground state, energy is emitted in the form of radiation in the visible region. The heat excites the valence electron to a higher energy level. When alkali metal salts are placed in a flame and reduced to their gaseous metal atoms, they emit characteristic colors. In this reaction, the hydrogen atom gains an electron and is present as the hydride ion.ĭue to their high reactivity, alkali metals oxidize easily upon exposure to air and are therefore stored in mineral oils. The heat produced during the reaction may even ignite the released hydrogen gas leading to fire or an explosion.Īlkali metals can also react with hydrogen to form alkali metal hydrides, often used as strong bases. The reaction becomes more violent for the heavier alkali metals given their lower ionization energies. The intensity of the reaction increases down the group as the ionization energies of the alkali metals decrease.Īlkali metals react vehemently with water to generate hydrogen gas and a solution of alkali metal hydroxide like potassium hydroxide, which is a base often used in soap making. The reaction of sodium metal with chlorine gas is highly exothermic and releases sparks and heat. This also explains why alkali metals are excellent reducing agents, with lithium being the least reactive and cesium the most.Īlkali metals react vigorously with nonmetals, such as halogens, to form salts. Their low ionization energies indicate that alkali metals readily lose electrons to become cations with a noble gas configuration. The electron configuration of these elements implies that the valence shell has only one electron. The alkali metals (group 1) have ns1 valence electron configurations and form M + ions, while the alkaline earth metals (group 2) have ns2 valence electron configurations and form M 2+ ions. Moving down the group, the mass, density, and atomic radii increase, while the melting points and ionization energies decrease. The chemical families consist of elements that have the same valence electron configuration and tend to have similar chemistry. Francium is radioactive and has no stable isotopes.Īll alkali metals are unusually soft and share metallic properties, such as high electrical and thermal conductivity. \): Periodic table by Dmitri Mendeleev, 1871.Alkali metals are group 1 elements, including lithium, sodium, potassium, rubidium, cesium, and francium. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
